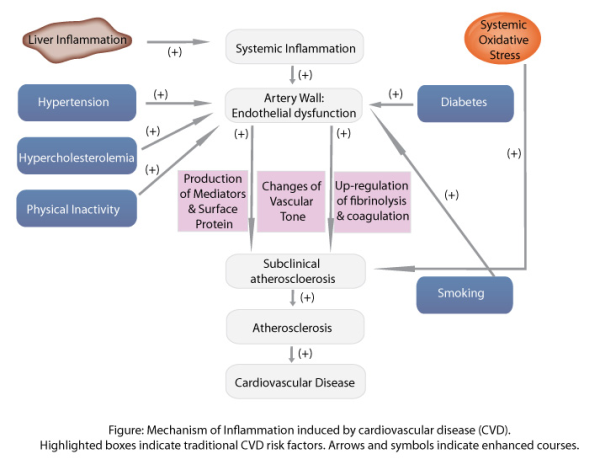
Rheumatoid arthritis (RA) is a chronic autoimmune joint disease characterized by inflammation of the synovium and destruction of cartilage and bone. During synovial inflammation, inflammatory cells (macrophages, mast cells, dentritic cells and lymphocytes) are recruited while resident cells (fibroblast synoviocytes, chondrocytes, osteoclasts, and osteoblasts) are altered to support the inflammatory process. Together, these events create a pathological tissue response.
The synovium consists of two layers, the sublining and intimal lining. In RA, the sublining becomes infiltrated with mononuclear cells, B lymphocytes produce autoantibodies, blood vessels proliferate, lymphoid aggregates form and the intimal lining shows increased cellularity. Macrophages in the synovium produce pro-inflammatory cytokines, chemokines and growth factors which in turn activate fibroblast-like synoviocytes (FLS) to produce their own array of mediators (e.g. proteolytic enzymes, chemokines and cytokines). This produces a paracrine/autocrine network that leads to synovitis, the recruitment of new cells and the destruction of the extracellular matrix. Fibroblast-like synoviocytes have emerged as key pro-inflammatory cells promoting the disease, largely due to their ability to produce massive amounts of degradative enzymes.
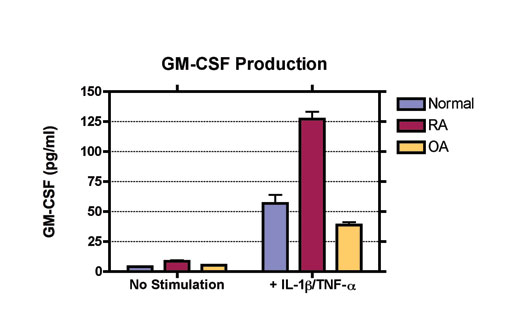
The availability of biological therapies has improved clinical outcomes by decreasing inflammation and joint destruction, however only about half of the patients exhibit substantial efficacy. Targeting FLS may further improve clinical outcomes without suppressing systemic immunity. In vitro FLS assays can be used to evaluate effective therapies for arthritis. Using FLS obtained from normal, RA and OA patients, we can evaluate a compound's effect on the production of pro-inflammatory mediators in a preclinical in vitro model.