MD Biosciences, a global biotechnology company focused in inflammations and neurology research, is expanding its line of arthritis and pain preclinical services with the addition of the CFA-induced monoarthritic pain model. The model offers advantages over traditional arthritis models in that disease develops in only one joint allowing a comparison to the healthy joint. The quantification of pain associated with arthritis can be useful in elucidating the mechanisms of arthritis as well as evaluating the efficacy of anti-arthritic drug compounds in the discovery phase.
The monoarthritic pain model is a 28-day model with arthritis and subsequent pain present in a single joint. Pain response develops 1 day post-induction. Readouts available are clinical signs and scoring, body weights, mechanical and thermal stimuli, weight bearing, histology and cytokine analysis.
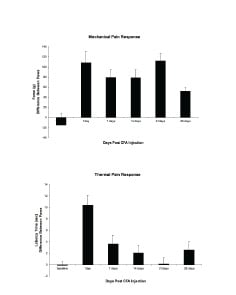
In addition to the monoarthritic pain model, MD Biosciences offers the traditional Adjuvant-induced arthritis model that allows for traditional pain measurements as well as a record of spontaneous pain using the Foot Print of Pain method. Other Arthritis models and research reagents include the Collagen Induced Arthritis (CIA) and Collagen Antibody Induced Arthritis (CAIA), ArthritoMab™ Arthritogenic Antibody Cocktail, Collagen Reagents, Collagen Antibodies and Collagen-related ELISA Kits and Assays.
About MD Biosciences
MD Biosciences provides products and pre-clinical services for companies engaged in inflammations and neurology research. The company is headquartered in Switzerland and has specialized laboratories located in the United States, United Kingdom, and Israel. A panel of scientific experts provides companies’ in-depth expertise and technologies to tackle problems and provide flexible drug discovery solutions, enabling smarter results faster.
###
The information in this press release should be considered accurate only as of the date of the release. MD Biosciences has no intention of updating and specifically disclaims any duty to update the information in these press releases.







