3 min read
By: MD Biosciences on Jun 25, 2012 3:20:00 PM

 Matrix metalloproteinases (MMPs) are zinc-dependent endopeptidases that function to degrade extracellular matrix. The turnover of ECM in organs is regulated by a balance by MMPs and their inhibitors (TIMPs) and the imbalance is implicated in a variety of diseases. Here we focus on the roles of MMPs in ischemia - particularly cerebral stroke and myocardial infarct.
Matrix metalloproteinases (MMPs) are zinc-dependent endopeptidases that function to degrade extracellular matrix. The turnover of ECM in organs is regulated by a balance by MMPs and their inhibitors (TIMPs) and the imbalance is implicated in a variety of diseases. Here we focus on the roles of MMPs in ischemia - particularly cerebral stroke and myocardial infarct.
Two MMP family members, MMP-2 and MMP-9, are known to degrade the major components of the BBB basal lamina (i.e. type IV collagen, laminin and fibronectin) and have been implicated in ischemic stroke. Inflammatory cytokines repidly up-regulate MMP-9 first in endothelial cells, then in neutrophils (which degranulate releasing pro- and active forms of MMP-9) and finally in macrophages. MMP-9 knock-out and neutrophil degranulation suppression strategies in preclinical cerebral ischemia models have been shown to reduce stroke damage. Futher, SB-3CT, a selective inhibitor of MMP-2 and MMP-9, appears to reduce lesion volume in preclinical models of stroke. Additionally, statin drugs and Minocycline both appear to have inhibitory effects on MMPs and are known to improve outcomes in preclinical cerebral stroke models. Researchers are focused on MMPs and how they contribute to the pathological events in the brain following stroke, traumatic brain injury and other neurodegenerative effects.
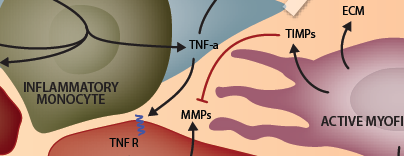
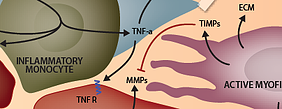
Early or exaggerated degradation of existing ECM or late/incorrect generation of new ECM can jeopardize the structural integrity of the heart leading to geometric changes in the LV that make it more susceptible to rupture, dilation, arrhythmias and pump failure. After myocardial infarct (MI), pro-inflammatory cytokines including TNFa and IL-1b act to increase both the expression and the activity of MMPs in cardiac tissues, resulting in proteolytic cleavage of a variety of ECM proteins. In addition to their prominent role in matrix degradation, MMPs also exacerbate the post-MI inflammatory environment in a number of ways. MMPs are responsible for post-translational processing of many cytokines, chemokines and growth factors - such as shedding them from membranes to create soluble forms, activating them by cleaving pro regions or making other modifications, many of which contribute to the promotion of inflammation. Low molecular weight ECM fragments generated by MMP proteolytic activity are detectable in circulation within minutes of myocardial infarct. This ECM debris has pro-inflammatory properties and is capable of eliciting leukocyte chemotaxis with some evidence sugging that collagen fragments may signal through CXCR1 and CXCR2 chemokine receptors on neutrophils and low-molecular with hyaluronan fragments may infuced pro-inflammatory cytokine expression in macrophages and endothelial cells via interactions with CD44. As inflammation resolves, anti-inflammatory cytokines, including TGFb, up-regulate expression of Tissue Inhibitor of Metalloroteinases (TIMPs) allowing newly-generated matrix to be preserved. A significant amount of experimental evidence from preclinical myocardial ischemia models suggests that MMP inhibitors may have cardioprotective effects (which has yet to be observed in the clinical setting).