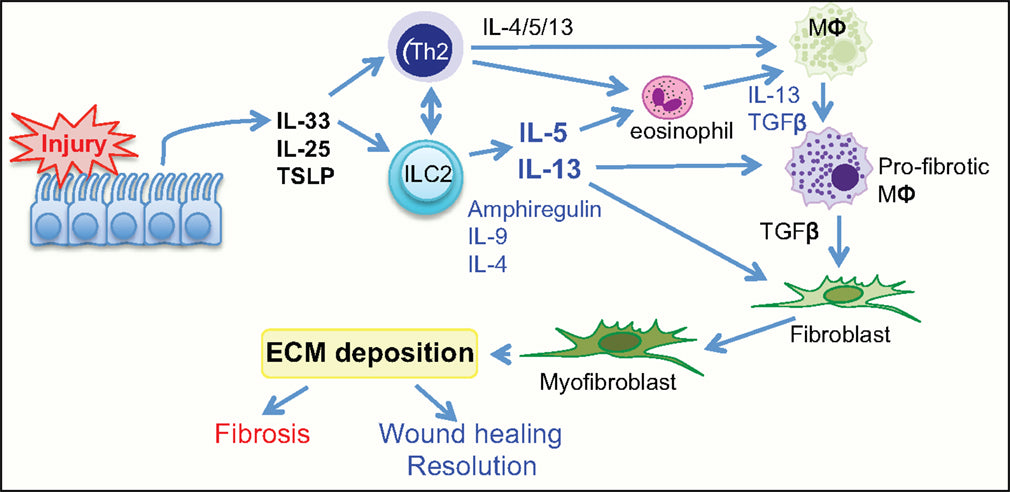
IL-33 and T1/ST2 function in wound healing and Inflammatory Responses
Aug 07 , 2008
IL-33 is a member of the IL-1 cytokine family with a widely-recognized inflammatory role in a broad range of skin diseases. It functions as a signaling ligand for the T1/ST2 receptor, also known as IL1RL1, T1 and IL-33R (Palmer and Gabav, 2011). Expression of IL-33 occurs in the nucleus of epithelial and endothelial cells and has been labeled as an “alarmin”—an immunological alarm signaled during cellular distress or cellular necrosis in mammals. Interestingly, recent reports indicate distinct species differences between mice, humans and pigs, prompting further comparative studies. In some murine epidermis models, prominent constitutive IL-33 expression was seen in epidermal keratinocytes and was drastically lost in basal keratinocytes during acute inflammatory events, while other studies observed a drastic increase in IL-33 during inflammatory events (Sundes et al., 2015). The loss of IL-33 from keratinocyte nuclei could be due to protein degradation or another release mechanism not yet identified. In contrast to the prominent constitutive IL-33 expression in murine keratinocytes, IL-33 signal was substantially low in human epidermis, demonstrating weak signals in the nuclei of keratinocytes. The classification as an alarmin is suitable for murine models but may not be as accurate for humans and pigs.
A primary function of IL-33 is to act as an immune response factor in keratinocytes playing a role in early wound healing responses in both pigs and humans. It is known to enhance wound healing via mucosal healing and epithelial restoration and repair. Researchers have shown that in steady state human wound healing models, IL-33 expression stayed weak in keratinocytes and IL-33 was down regulated in endothelial cells by pro-inflammatory cytokines (Sundes et al., 2015). Despite little to no expression of IL-33 in the steady state in pig and human keratinocytes, there was an upregulation 24 hours after wound creation. In an ex vivo organ culture, exposing human skin to IFN-γ caused an upregulation of IL-33, but it had no effect on IL-33 levels in mouse skin. Thus, keratinocyte-derived IL-33 is an inflammation-induced factor and not a constitutively expressed alarmin in the human epidermis. Similarly, a pig skin wound model indicated that non-inflamed skin exhibited strong endothelial signals for IL-33 but no detectable expression of IL-33 in the epidermis. Moreover, a clear induction of IL-33 in keratinocytes was observed at the edge of the wound in pigs, mimicking human skin regulatory patterns (Sullivan et al., 2001; Dawson et al., 2013).
An explanation for these significant species differences between mouse, pig and human models is that the immune system develops in response to environmental conditions. IL-33 is recognized as a soluble cytokine, but its function could reflect the functioning of an endogenous, nuclear factor (Ogryzko et al., 2014). While IL-33 is only found in mammals, IL1RL1 gene coding for ST2, is also present in non-mammals such as zebrafish (Geer et al., 2010). This could indicate that ST2 developed a ligand-binding capacity at a subsequent evolutionary stage, whereas IL-33 might have had its evolutionary origin as a nuclear factor prior to emerging as a ligand. Another possibility is attributed to distinct intracellular functioning since IL-33 can have opposite effects. In recent lesion models, there were some IL-33-independent and IL-33-dependent developments, reiterating the need for further exploration into expression patterns (Salimi et al., 2013). With IL-33 being a multi-factorial cytokine demonstrating a versatile role in wound healing, inflammatory processes and autoimmunity, understanding underlying mechanisms of ST1/ST2 signaling pathways is valuable in strategizing novel developments for wound healing and inflammatory diseases.
Products for T1/ST2 Research:
T1/ST2 Mouse Monoclonal Antibody
T1/ST2 Monoclonal Antibody, FITC-conjugated
T1/ST2 Monoclonal Antibody, Biotinylated
T1/ST2 Monoclonal Antibody, PE-Conjugated
T1/ST2 Monoclonal Antibody, azide-free
ST2L Monoclonal Antibody, FITC-conjugated
ST2L Monoclonal Antibody, Biotinylated
Image source: https://www.frontiersin.org/articles/10.3389/fimmu.2015.00597/full
