Evaluating neuroprotective therapies for Parkinson's Disease
A “classical” animal model of PD is created by the systemic administration of MPTP, a highly lipophilic compound that readily crosses the blood-brain barrier. This mouse model is the most commonly used PD animal model, and is similar to the 6-OHDA model with respect to how the MPTP model shows selective degeneration of DAergic neurons. Interestingly, rats are relatively resistant to MPTP and thus do not lend themselves for MPTP exposure to produce a PD animal model (4). The other major benefit of this mouse model is that MPTP is known to cause a PD-like disease in humans by a similar mechanism of action (3). Unlike 6-OHDA, though, MPTP itself is not an active neurotoxin. Rather, following systemic injection (typically intraperitoneally) it is taken up by glial cells (e.g. astrocytes), gets converted to 1-m- ethyl-4-phenyl-2,3-dihydropyridium (MPDP) by monoamine oxidase B (MAO B) and is rapidly oxidized to the active form 1-methyl-4-phenylpyridinium (MPP+) (5). Upon release into the extracellular space, MPP+ is preferentially taken up by DAergic neurons through DAT and most severely affects the SN. Similarly to 6-OHDA, MPP+ kills neurons by inhibiting complex I of the mitochondrial respiratory chain. As a result of these neurotoxic effects, MPTP-injected animals also display motor abnormalities.
Assessments:
Don't see the model or assessment you require? Our scientists are experienced with rapidly validating models from the literature. Please speak with a scientist to discuss your requirements.
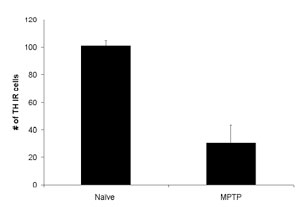
Total count of Th-IR immunoreactive cells in naive and MPTP-induced animals.
- Biotechnology company. East coast, United States