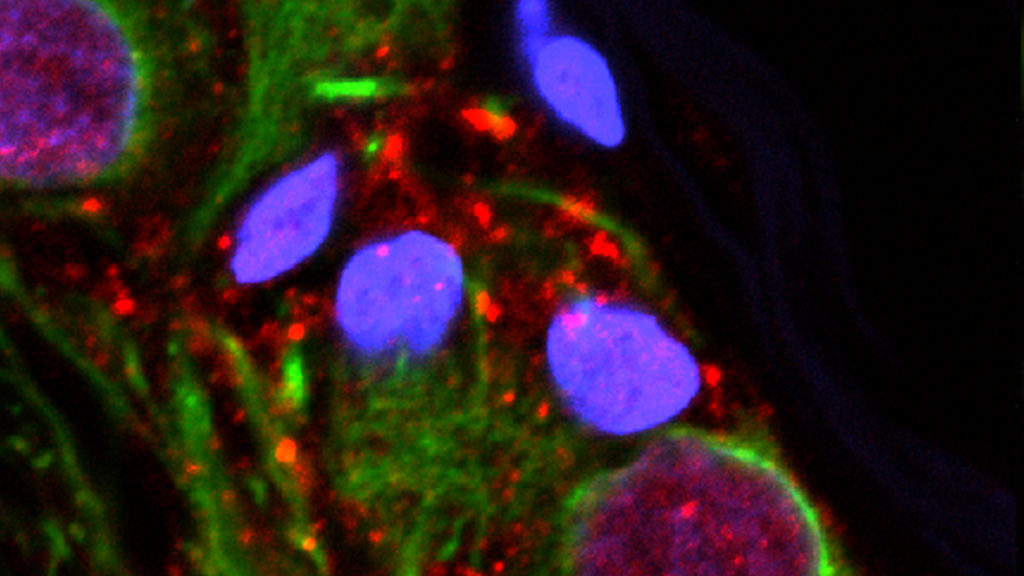
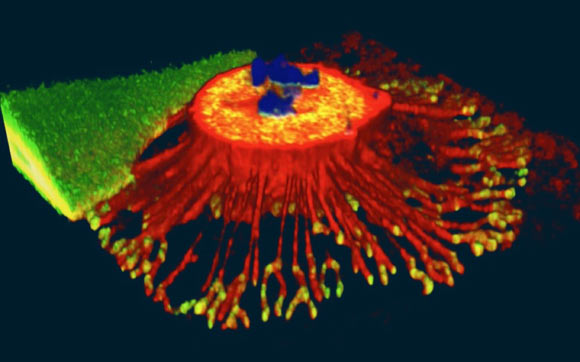
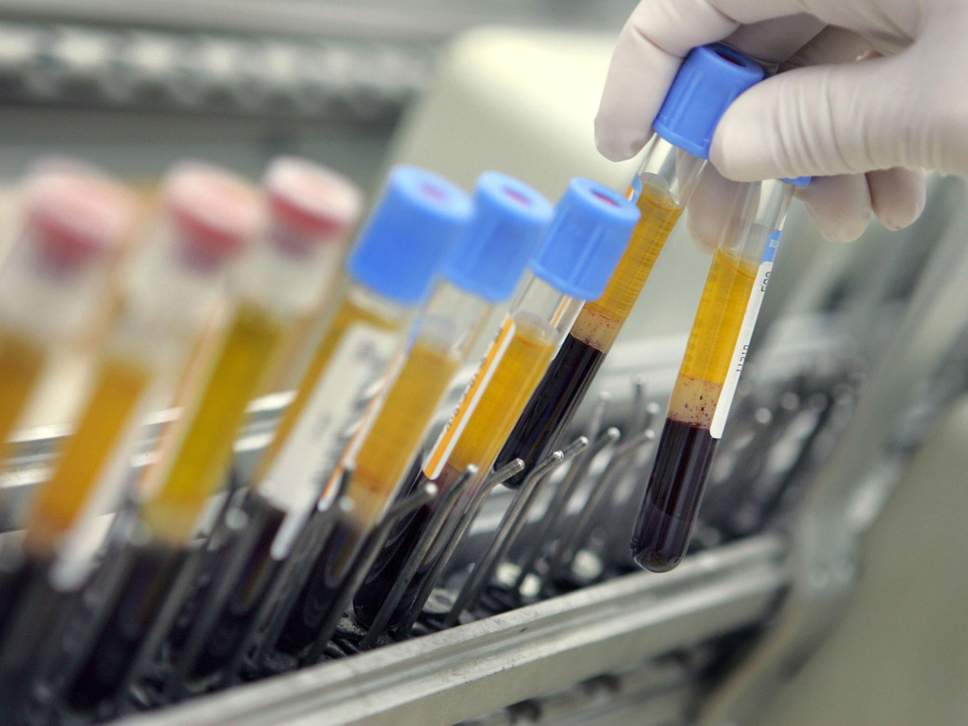
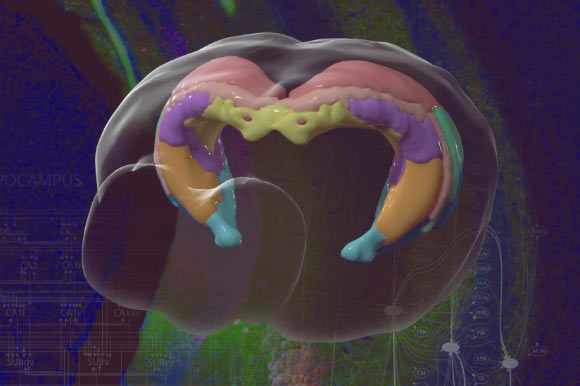
MD Biosciences providing up-to-date news coverage compiling posts from science journals including British Journal of Cancer, Proceedings of the National Academy of Sciences, Neuroscience News, ScienceDaily, Science Translational Medicine and Stat News, reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Neuro/CNS,
cancer/tumor,
Biomarkers,
Academic Research,
Immuno-Oncology,
Biotechnology,
Neuroscience
MD Biosciences providing up-to-date news coverage compiling posts from science journals including Nature Cell Biology, Sci News, Journal of PLOS Biology, Medical Press, ScienceDaily and PhysOrg, reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Neuro/CNS,
cancer/tumor,
Biomarkers,
Academic Research,
Immuno-Oncology,
Biotechnology,
Neuroscience
MD Biosciences providing up-to-date news coverage compiling posts from science journals including BioSpace, Jama Neurology, New England Journal of Medicine, Nature Communications and Science reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Neuro/CNS,
Biomarkers,
Academic Research,
Biotechnology
MD Biosciences providing up-to-date news coverage compiling posts from science journals including Nature Neuroscience, News: Medical and Life Sciences, Nature, Science Translational Medicine and Sci News reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Neuro/CNS,
Biomarkers,
Academic Research,
Biotechnology
MD Biosciences providing up-to-date news coverage compiling posts from science journals including Cell Death and Disease, Cell Press, Journal of Craniofacial Surgery, Nature Medicine, ScienceDaily, Science Translational Medicine and Sci News reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Neuro/CNS,
Biomarkers,
Academic Research,
Biotechnology
MD Biosciences providing up-to-date news coverage compiling posts from science journals including Cancer Research, Sci News, Nature Genetics, British Journal of Pharmacology and Science reporting fascinating and innovative discoveries happening all over the world across a wide variety of disciplinary areas. Skim the titles below to catch up on what is happening and to learn what the team at MD Biosciences is reading about. Follow the links to read more about something that catches your eye!
Read More
Topics:
Inflammation,
Neuro/CNS,
cancer/tumor,
dermatitis,
preclinical research,
Biotechnology
As we wind down to years end, we at MD Biosciences would like to thank everyone, especially our collaborators, for making this year a success. We have undergone significant growth that we expect to continue throughout the upcoming year.
Read More
Topics:
Pain,
Inflammation,
Neuro/CNS,
post-operative pain,
cancer/tumor,
Microbiome,
Biomarkers,
CLIA,
Neuropathy
Preclinical stroke models are critical to our understanding of the mechanisms and neurological deficits following human stroke. While reducing infarct size is a focus of stroke therapies, much attention is also on neuroprotective properties. Adding behavioral and functional outcome measures to preclinical studies is important to evaluate the impact on impairments that occur following stroke: learning, memory, motor function and sensory. There are many behavior tests, each having different sensitivities to deficits associated with particular areas of brain damage.
Read More
Topics:
Neuro/CNS
There are currently a large number of well-characterized, ischemic stroke animal models available for pre-clinical research. These models can be categorized into those two groups – those for the study of stroke-associated risk factors and those for the study of stroke pathophysiology. The latter can be further separated into models of focal verses global ischemia and are listed:[1]
Read More
Topics:
Neuro/CNS